Non-Invasive Endometrial Receptivity Testing
How does non-invasive receptivity testing work?
Endometrial receptivity testing can be a powerful tool for certain patients, but until now has required a costly and time-consuming mock cycle, followed by an invasive and uncomfortable endometrial biopsy.
Inti Labs’ research into microRNA biomarkers as alternatives to mRNA has paved the way for a non-invasive testing solution that leverages stable biomarkers in the bloodstream to accurately assess a patient’s endometrial status, with the additional possibility of avoiding a mock cycle for sample collection purposes.
Why microRNA?
Endometrial receptivity tests were originally performed using endometrial tissue samples. microRNA (miRNA) biomarkers in the bloodstream can identify those same endometrial conditions.
These miRNAs in the bloodstream have been found to regulate a number of functions during pregnancy, including glandular development, responses to oxidative stress, and cell cycling. Through the regulation of these mechanisms, miRNAs can affect the endometrium’s environment, growth process, and, by extension, pregnancy results.
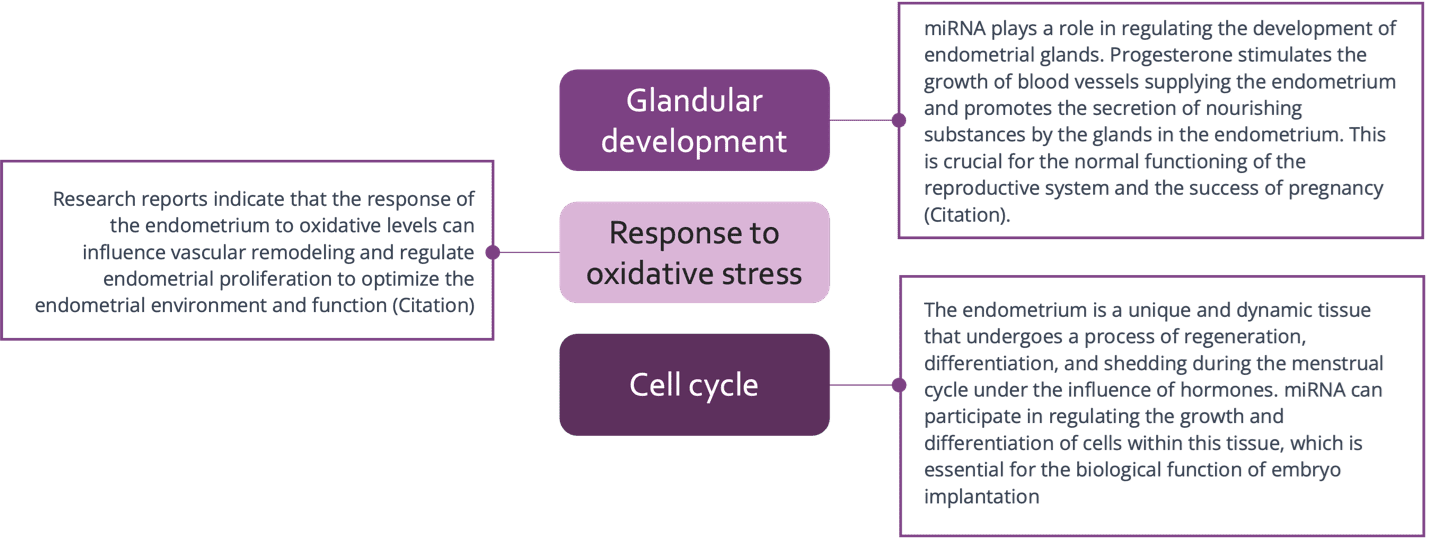

Inti Labs was able to identify blood-based miRNA biomarkers that accurately reflect the endometrium’s status, similar to the traditional method of analyzing endometrial tissue samples, but without the invasive biopsy needed for sample collection.
ora™ uses these miRNA biomarkers to analyze the endometrium’s status, providing doctors with a more precise recommendation for the optimal time for embryo transfer.
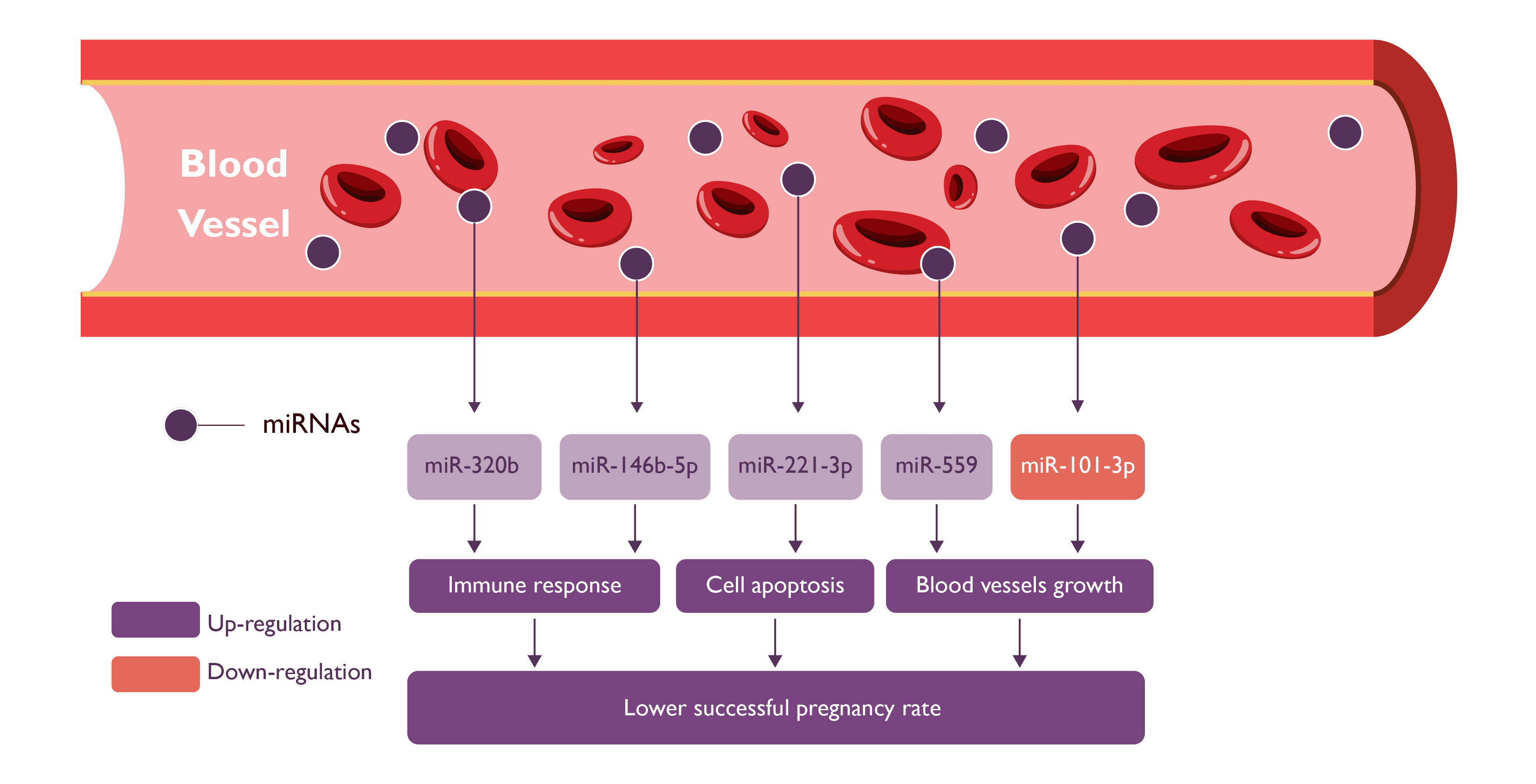

Comparing mRNA and microRNA-based tests
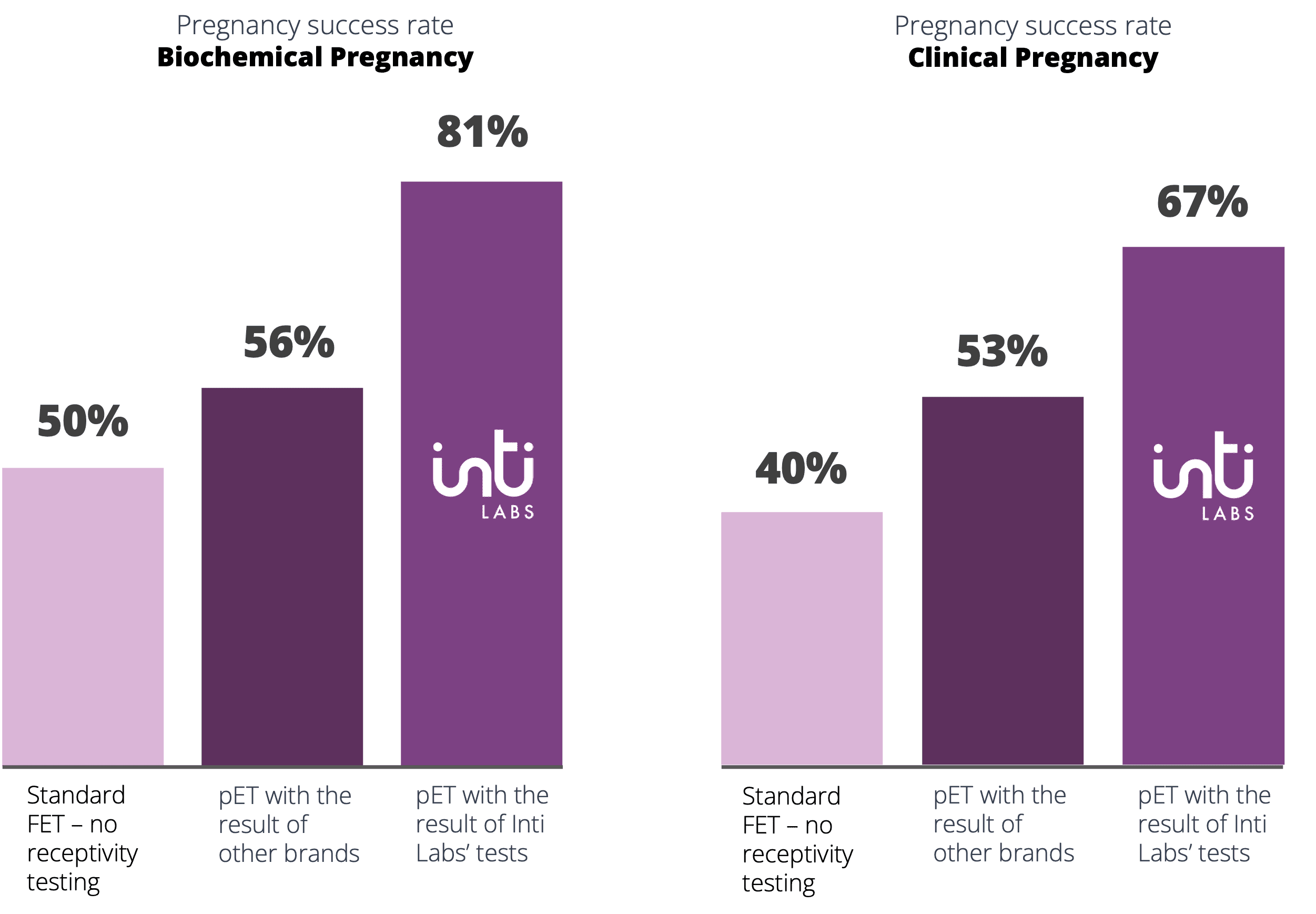
In both our first-generation product, MIRA™, and our non-invasive solution, ora™, we’ve found microRNA biomarkers to be more stable than those in other existing solutions. We’ve seen higher pregnancy rates when compared to patients that underwent competing tests.

What is ora™?
ora™ is the first and only non-invasive endometrial receptivity test for determining the optimal time for embryo transfer.
ora™ assesses over 300 microRNA (miRNA) biomarkers in the bloodstream that target over 700 endometrial receptivity-related genes, making ora™ the most comprehensive and accessible solution for identifying a patient’s personalized window of implantation (WOI).
Why choose ora™
Higher Pregnancy Rates
Clinics using our microRNA-based receptivity tests have consistently higher successful pregnancy rates when compared to those who use a competing test or do not undergo any form of receptivity testing. Use of ora™ has demonstrated that it can increase a patient’s chance of success to over 70%.
Improved Patient Experience
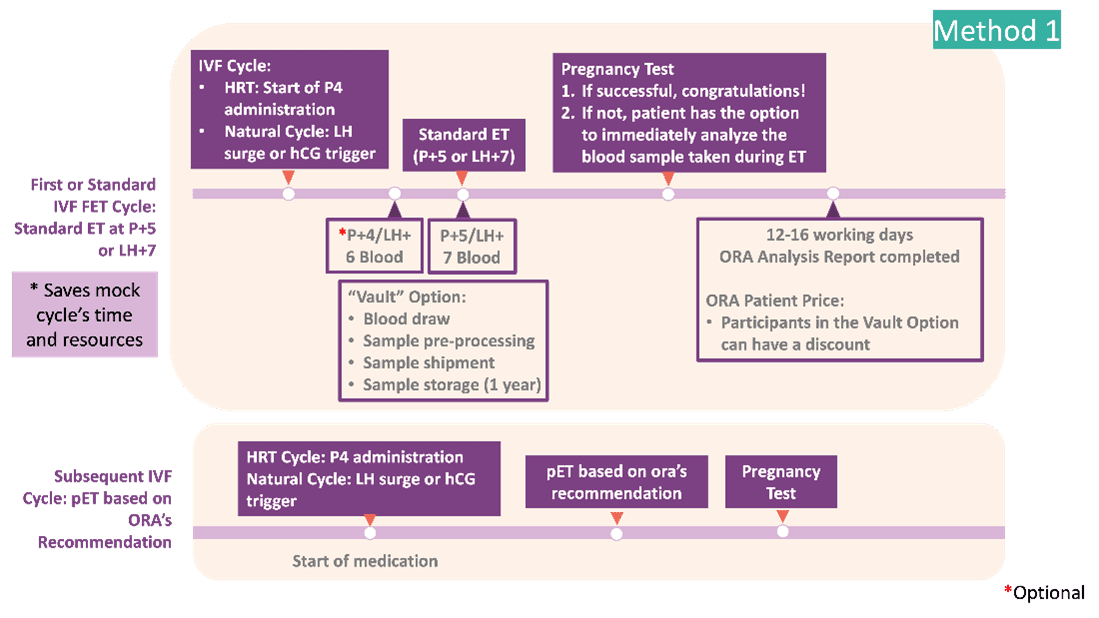
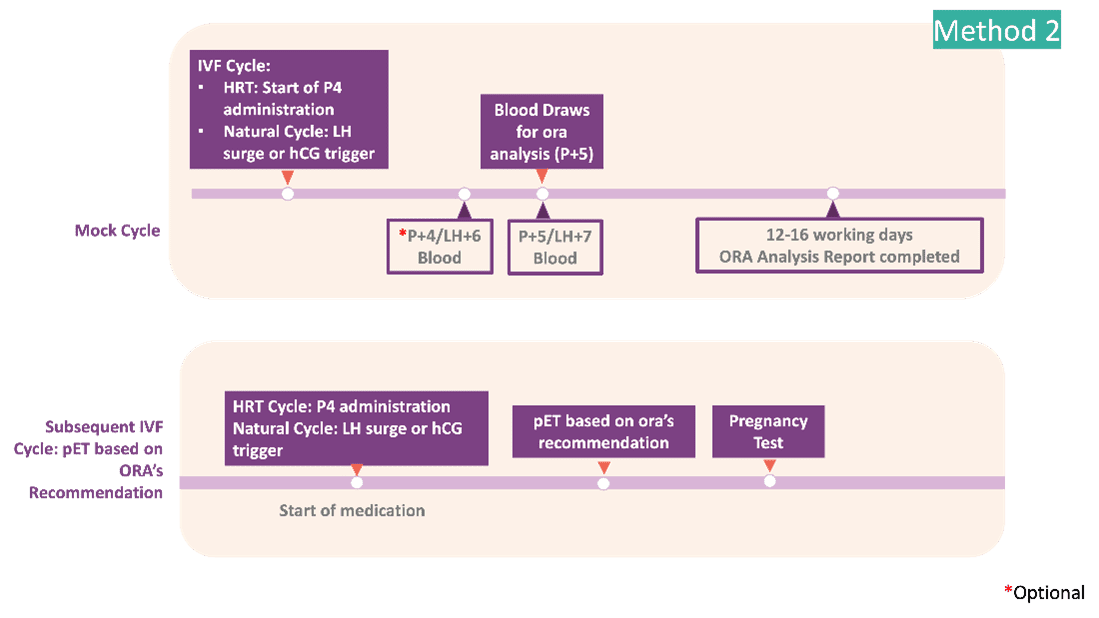
ora™ eliminates the need for an endometrial biopsy procedure. All that is required to accurately identify the patient’s WOI is a simple blood sample, taken on a standard embryo transfer day, or on day 5 of progesterone administration during a mock cycle.
Skip The Mock Cycle
When implemented early in a patient’s treatment plan, our Vault program bypasses the need for a costly and time-consuming mock cycle. Simply acquire a blood sample on the day of the patient’s initial FET, and we’ll store it until needed. If the implantation is successful, no further action is required. If implantation fails, we will analyze the sample to inform timing of the next treatment cycle.
Reduced Re-Testing Rate
In some cases of a post-receptive result, a patient might have a shortened window of implantation, which a single analysis cannot account for. Due to the non-invasive nature of ora™, a second blood sample can be taken on Day 4 of an HRT cycle or Day 6 of a natural cycle to account for this chance. In the case that ora™’s analysis suggests the possibility of a shortened window, the second blood sample can be utilized to confirm these findings.
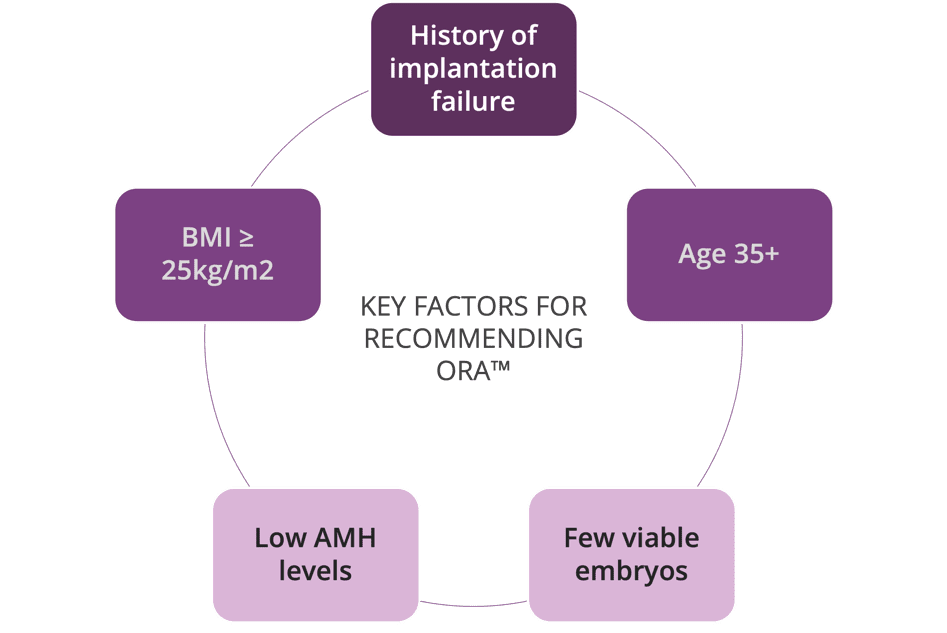
Which patients can benefit from ora™?
ora™ can accurately identify the WOI of any patient, but not every patient needs receptivity testing. We recommended ora™ for those who meet any of the following conditions:
- A history of implantation failure
- Few remaining high-quality embryos
- A lower- or higher-than-average BMI
- Age 35 or older
- Low AMH levels
The likelihood of having a displaced WOI increases with the number of implantation failures. Higher successful pregnancy rates have been seen after identification of the patient’s WOI.

How do I incorporate ora™ into the treatment process?
Due to the non-invasive nature of ora™’s blood-based test, the sample can be taken on the day of embryo transfer of a standard cycle (Day 5 of an HRT cycle or Day 7 of a natural cycle) and stored at our lab.
If the implantation is successful, no further action is required. If unsuccessful, we’ll analyze the blood sample so you can go straight into a new IVF cycle with adjusted timing.


What is the procedure for ora™ testing?
1. Complete the Sample Submission and Consent Form
Fill out the Sample Submission and Consent Form with your patient (included in the kit).
2. Schedule the Blood Draws
Schedule the blood draws based on either natural cycles or hormone replacement therapy cycles*.
3. Obtain the Blood Samples
Use the blood sample collection tubes provided in the kit. Invert the tube 5-10 times immediately after the blood sample has been collected.
4. Process the Blood Samples
The blood samples will be processed into plasma samples onsite within two hours of sample collection according to ora™’s laboratory instructions.
5. Store the Sample
Label the processed blood samples (plasma samples) with the unique sample barcode provided in the kit. The sample can be stored at room temperature.
6. Ship the Sample
Ship the package to the designated shipping address using priority or next day shipping.
“One of our patients had experienced several failed implantations and was down to her last viable embryo. After using ora™️, we received a Pre-Receptive result, meaning her window of implantation was 24 hours later than expected. Following ora™️’s recommendation for adjusted timing, her final embryo transfer and subsequent implantation was successful and is an ongoing healthy pregnancy.
The process was simple and patient-friendly — taking a blood sample is so much easier than an endometrial sample — and the benefits of a smoother and more comfortable experience for patients cannot be overstated. We look forward to continue offering our RIF patients a renewed chance at parenthood with the critical insights ora™️ provides.”
Dr. Tran Nguyen
Lyell McEwin Hospital, Embrace Fertility, FBW Gynaecology Plus
Adelaide, AUS
Want to learn more about ora™?
Download our in-depth overview of ora™, or visit our contact page to get in touch directly.
References
Chen, C. H., Lu, F., Yang, W. J., Yang, P. E., Chen, W. M., Kang, S. T., Huang, Y. S., Kao, Y. C., Feng, C. T., Chang, P. C., Wang, T., Hsieh, C. A., Lin, Y. C., Jen Huang, J. Y., & Wang, L. H. (2021). A novel platform for discovery of differentially expressed microRNAs in patients with repeated implantation failure. Fertility and sterility, 116(1), 181–188.
Click here for more info.
Chen M-J, Hsu A, Lin P-Y, Chen Y-L, Wu K-W, Chen K-C, Wang T, Yi Y-C, Kung H-F, Chang J-C, et al. Development of a Predictive Model for Optimization of Embryo Transfer Timing Using Blood-Based microRNA Expression Profile. International Journal of Molecular Sciences. 2024; 25(1):76.
Click here for more info.